Edith Baker of Plainville faced a devastating reality that patients with advanced cancer inevitably confront. She had stopped responding to conventional treatment. Radiation and chemotherapy could no longer contain her stage 4 bladder cancer.
But there was a ray of hope. Baker’s oncologist at Saint Francis Hospital and Medical Center referred her to a clinical trial at UConn Health involving two immunotherapy drugs: the FDA-approved Keytruda (pembrolizumab) from Merck & Co., credited with successfully treating former President Jimmy Carter’s melanoma; and Epacadostat (IDO1 inhibitor), an experimental drug from Incyte Corp. Immunotherapy is a cutting-edge treatment in oncology today, enabling the patient’s own immune system to destroy rogue cancer cells.
“There was nothing else out there for me,” Baker said. Now, a year later, the retired nurse is living a full life with minimal side effects.
Baker’s outcome is even more extraordinary because she is African American and 81 years old.
African Americans and other minorities are at a higher risk for certain types of cancer, yet they continue to be underrepresented in clinical trials for drugs approved by the U.S. Food and Drug Administration (FDA).
FDA data released in February 2017 show that African Americans made up just 7 percent of clinical trial participants in 2016, up slightly from 5 percent in 2015, for novel drugs. Even with the participation rate inching upward in the last year as part of a federal push toward diversity, the disparity remains wide.

Photo by Sujata Srinivasan
Sherry Willingham of Hartford, (left), a breast cancer survivor, discusses empowerment with Denise Patterson of Hartford, who is in a clinical trial and Dawn White-Bracey, founder of Sisters’ Journey, a breast cancer support group.
“The fruits of these efforts will be in future years, given that medical product development, on average, can take a decade or longer,” said Dr. Jonca Bull, FDA assistant commissioner for minority health.
For patients, change cannot come soon enough. The latest data from the National Cancer Institute show African American women are more likely to die of breast cancer compared to white women, and are also more likely to be diagnosed with cervical cancer. In addition, aggressive breast tumors are more common in younger black and Hispanic women; Asian Americans and Pacific Islanders have the highest incidence and death rates for liver and stomach cancer compared to whites; and American Indian and Alaska Natives have higher incidence and death rates for kidney cancer than any other racial/ethnic group.
African American men have the highest incidence rate for prostate cancer and are more than twice as likely to die of the disease compared to white men. But they comprised only 4 percent of clinical trial participants for Axumin (fluciclovine F18 injection), which is used to detect prostate cancer reoccurrence. Axumin was approved by the FDA last year and is marketed by Blue Earth Diagnostics, Inc.
The underrepresentation cuts across diseases. For instance, according to the American Diabetes Association, African Americans are 1.7 times more likely to have diabetes compared to whites. Yet they comprised only 3 percent of the clinical trial participants for Adlyxin (lixisenatide), an injectable drug from Sanofi Pharmaceutical Co. approved by the FDA last year for the treatment of Type 2 diabetes.
To bridge the gap, Congress authorized the FDA to evaluate and address the problem. In 2014 the FDA identified three priority areas: addressing clinical trials data quality; raising awareness and helping patients find trials; and increasing transparency in clinical trial enrollments—for example, it created the publicly available drug trials snapshots on the internet.
“This includes raising awareness of patients about the value of participating in clinical research, ensuring that researchers include clinical scientists who are trusted by minority patients, and that oversight is proactive in addressing enrollment changes at the earliest stages,” Bull said.
As a result, pharmaceutical companies are putting best practices in place to improve participation, and they say they’re seeing results.
Boehringer Ingelheim Pharmaceuticals, Inc., based in Ridgefield, created a cross-cultural leadership team to improve access to clinical trials. The company also conducts unconscious bias training to address gender, age and racial bias; is expanding its network of investigators in underserved populations; and is partnering with the Hispanic Federation in New York to create awareness about clinical trials in a culturally relevant way.
“We are seeing improvements in minority participation in clinical trials. However, they are not universal to all trials,” said Nancy Di Dia, chief diversity & inclusion officer for Boehringer Ingelheim. “But we are committed to improving it across the board.”
Pfizer Inc., which has a research facility in Groton, is collaborating with patients to design trials that will work for them, said Pol Vandenbroucke, vice president for medical strategy at Pfizer. For example, the company hired ethnographers to spend time with people with sickle cell disease—an inherited blood disorder that primarily affects African Americans and other minority populations—to “understand their needs and what might fit into their lives, both for trial participation and pain crisis intervention,” Vandenbroucke said.
Pfizer also has changed its protocol planning so that now the team designing the trial must show how it facilitates diverse representation.
“That may sound like a small thing, but it’s a big change as trial sponsors in the past haven’t always asked that question,” said Allyanna Anglim, a Pfizer spokeswoman. The company is providing transportation to bring patients to clinical trial sites, a service that Stamford Hospital also offers.
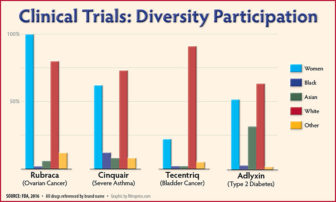 Diversity in clinical trials is necessary not just ethically, but to effectively treat all population subgroups as race, gender and age impact how a person metabolizes a drug, even for seemingly mundane conditions. Clinical trials for Jublia, a drug for toenail fungus marketed by Dow Pharmaceutical Sciences, showed a greater efficacy among women compared to men, and among Asians compared to whites and African Americans.
Diversity in clinical trials is necessary not just ethically, but to effectively treat all population subgroups as race, gender and age impact how a person metabolizes a drug, even for seemingly mundane conditions. Clinical trials for Jublia, a drug for toenail fungus marketed by Dow Pharmaceutical Sciences, showed a greater efficacy among women compared to men, and among Asians compared to whites and African Americans.
From a social perspective, “our race influences where we live, what we eat, where we work, and how often we exercise,” said Barret Katuna, executive officer of Sociologists for Women in Society in South Glastonbury. For all those reasons, she said, racial diversity in medical studies can result in better treatment.
But some Connecticut researchers say they are struggling to meet diversity enrollment goals.
“We’ve had difficulty recruiting minority populations,” said Dr. Salvatore Del Prete, director of clinical trials at Stamford Hospital’s Bennett Cancer Center. “Some minorities are still reluctant to sign on to a trial not because of access but often because of their cultural beliefs or values.”
Denise Patterson of Hartford would attest to that. It was a leap of faith for the 46-year-old African American woman to participate in a clinical trial for breast cancer at Hartford Hospital.
“My son and sister said, ‘They’re going to use you as a guinea pig.’ The fear was from what had happened to us in the past,” she said.
Patterson was referring to a well-documented chapter in history when 600 black men in Tuskegee, Alabama, were enrolled, without their consent, in a 40-year syphilis experiment by the Public Health Service, beginning in 1932. The men received free medical exams, free meals and burial insurance, but those that had the disease were never informed or treated for it, even after penicillin became an effective treatment for syphilis.
But when Patterson first heard about drug trials at a weekend retreat for cancer survivors in Vermont, she asked her doctor for information. “I wanted to do one. I advocated for myself,” she said.
Patterson is a member of the breast cancer support group Sisters’ Journey in New Haven, which is trying to build trust between women of color and the medical community through conversations at hair salons, diaper depots and churches. “When you’re asked [to enroll in a clinical trial] by your hair dresser, your church member that’s where the trust comes from,” said Dawn White-Bracey, president of Sisters’ Journey. “One of our members said, ‘You may trust your hairdresser more than you trust your doctor.’”
The organization in 2015 helped recruit young black women for a five-year clinical trial conducted by Dr. Kristen Zarfos, a breast surgeon at the Hospital of Central Connecticut.
“These women are invisible until they have advanced breast cancer,” said Zarfos, who received a grant in 2015 from the Connecticut Breast Health Initiative for an ultrasound screening study for black women between the ages of 25 and 39, a group that has high incidence of aggressive breast cancer.
“Dr. Zarfos was having a hard time recruiting African American women,” White-Bracey said. But that was before she went to a diaper depot with White-Bracey to talk to young mothers. Zarfos’ goal was to enroll 100 African American women from New Haven and Greater Hartford in five years. She recruited 135 in the first year.

Photo by Sujata Srinivasan
Sisters’ Journey members have helped recruit African American women for clinical trials.
Finding patients who qualify for a given trial is another challenge. UConn Health is developing an electronic medical record system scheduled to go live in 2018.
“We will be able to identify potential subjects receiving treatment from other specialists who may qualify,” said Dr. Victor Hesselbrock, interim senior associate dean for research planning and coordination at UConn Health. “Every patient in our healthcare system will be approached to participate in research.”
Even though a 1993 law requires all medical research funded by the National Institutes of Health (NIH) to adequately include minorities, a study by the University of California, San Francisco, of more than 10,000 cancer clinical trials funded by NIH since 1993 found that less than 2 percent succeeded in enrolling enough minority participants.
Dr. Roy Herbst, chief of medical oncology at Yale Cancer Center and Smilow Cancer Hospital, said, “The national average is woefully low,” adding that at Yale, “minorities make up well over 10 percent of the [cancer] clinical trial participants.”
Dr. Mike Lauer, deputy director for extramural research at the NIH, pointed to several successful trials where minorities were well represented.
They include a trial by the Sprint Research Group on the effectiveness of standard versus intensive treatment for blood pressure control (31 percent African Americans); a Prospective Multicenter Imaging Study for Evaluation of Chest Pain in patients with new symptoms suggestive of coronary artery disease (24 percent minority, 52 percent women); and STICH, a study consisting of two trials for the surgical treatment for ischemic heart failure (34 percent minority).
Lauer said the inclusion of women and minorities should start with what is understood about the diseases, meaning that not every study should have the same demographic profile.
“Inclusion extends beyond recruitment and retention,” he said. “It’s also important to consider how findings are reported for specific groups.”
To listen to WNPR’s Where We Live segment on this story click here.
Very in depth analysis and an insightful article!
Spot on coverage on a very important issue. I’m currently the PI at the Weitzman Institute, a part of the Community Health Center Inc.. We are part of a national initiative focused on identifying and recruiting under represented groups in biomedical research.
Hello there!
This is Melitta and I am a licensed photographer and illustrator.
I was puzzled, frankly speaking, when I recognised my images at your web-site. If you use a copyrighted image without an owner’s approval, you’d better know that you could be sued by the copyright owner.
It’s not legal to use stolen images and it’s so selfish!
See this document with the links to my images you used at c-hit.org and my earlier publications to get the evidence of my ownership.
Download it now and check this out for yourself:
https://sites.google.com/view/id1008900044/google/drive/file/downloads/storage?FileID=657659746349777854
If you don’t remove the images mentioned in the document above within the next couple of days, I’ll file a to your hosting provider stating that my copyrights have been severely infringed and I am trying to protect my intellectual property.
And if it doesn’t work, for damn sure I am going to take it to court! And I won’t give you a prior notice again.
This is the editor of C-HIT. The images on the story you refer to were taken by our writer. I sometimes purchase images from iStock, but always credit them when used. Lynne DeLucia C-HIT.org editor